Orthogon Therapeutics today announced the closing of a follow-on $11 million financing, bringing its total capital raised to $36 million. This financing supports the continued advancement of its first-in-class drug against BK polyomavirus. BK virus infections are a major cause of complications in transplant patients, with no approved treatments.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260417911548/en/
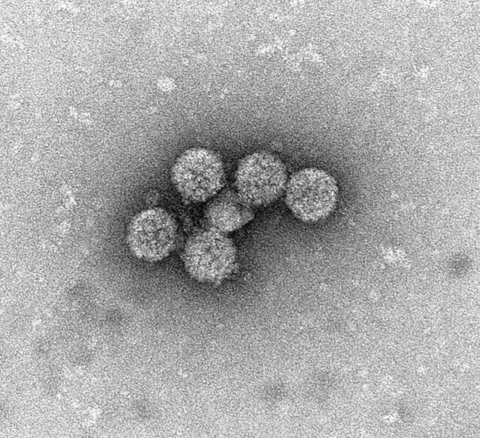
Electron microscope image of BK virus, showing the VP1 capsid protein that creates an icosahedral shell around the viral genome.
The company is pioneering an oral therapy that addresses the full spectrum of BK infection, from early reactivation through systemic spread and onset of severe disease. By targeting viral proteins previously considered inaccessible to small molecule drugs, Orthogon is advancing a solution where other therapeutic modalities have fallen short.
Orthogon’s lead asset uniquely targets the viral capsid protein (VP1), delivering potent antiviral effect at the site of viral replication. The intracellular activity leads to sustained control of viral infection across BK variants and related human polyomaviruses, particularly in transplant patients, where viral persistence drives disease.
“This is not a conventional antiviral setting. We built this program to meet the realities of transplant care,” said Ali H. Munawar, Ph.D., CEO of Orthogon Therapeutics. “These patients are treated within a narrow balance of immunosuppression, organ function, and high pill burden. We designed around those constraints, arriving at a candidate profile that we’re excited to take into development.”
In parallel, Orthogon has published findings examining hundreds of patient-derived BK virus sequences, showing that the virus carries pre-existing diversity at antibody-binding regions and that it replicates beyond the reach of circulating antibodies. These studies explain the limited clinical benefit observed with neutralizing antibodies, challenges that Orthogon’s drug is designed to overcome.
The program draws on Orthogon’s portfolio of novel small molecules directed at each of the two viral proteins: the VP1 capsid and large T antigen (LTAg), a capability that has eluded the field for decades. The program will be featured at leading transplant and virology meetings in 2026, building on findings presented at the ASN in 2025.
Alongside its core focus on polyomaviruses, Orthogon is advancing programs in additional areas of unmet need in transplant-associated infections.
About BK and polyomaviruses:
BK virus (BKV) is among the most widespread chronic viral infections in humans. A member of the polyomavirus family, BKV establishes a lifelong infection in 80–90% of healthy adults worldwide. Reactivation occurs in the kidneys of nearly half of all solid organ and stem cell transplant recipients, leading to severe complications and graft loss. Other human polyomaviruses, including JC virus and Merkel cell polyomavirus, cause fatal progressive multifocal leukoencephalopathy (PML) and aggressive Merkel cell carcinoma, respectively.
About Orthogon Therapeutics:
Orthogon is a polyomavirus-focused biotech built on a proprietary discovery platform that fuses structure-based drug design with deep biophysical interrogation of viral proteins, unlocking targets long considered undruggable. The company is headquartered in Greater Boston with a research branch in Leuven, Belgium. To learn more visit www.orthogontherapeutics.com
Orthogon Therapeutics LLC is an independent, privately held research & development (R&D) company affiliated with the Pledge Therapeutics discovery engine. More info on www.pledge-tx.com
View source version on businesswire.com: https://www.businesswire.com/news/home/20260417911548/en/
Media gallery
